 The Cs+ ion occupies the cubic hole in the center of a cube of Cl ions. Figure 12.9: The Cesium Chloride Structure. 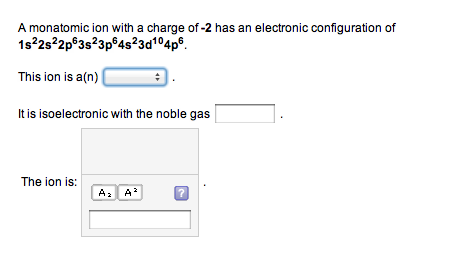
A type II ionic compound contains a metal that forms more than one type of ion, i.e., the same element in different oxidation states. The cesium chloride structure is most common for ionic substances with relatively large cations, in which the ratio of the radius of the cation to the radius of the anion is in the range shown in Table 12.2. These elements usually form ionic compounds. For example, calcium carbonate consists of the monatomic cation Ca 2+ and the polyatomic anion CO 2−ģ both pentazenium ( N + 5) and azide ( N − 3) are polyatomic as well.Ī type I binary ionic compound contains a metal that forms only one type of ion. Transition (B-group) and Post-Transition (Group IVA and VA) Metals. If, instead of being monatomic, an ion contains more than one atom, even if these are of the same element, it is called a polyatomic ion. Monatomic ions are formed by the gain or loss of electrons to the valence shell (the outer-most electron shell) in an atom. The resultant ion is symbolized as P-3 and is named the. Lewis Structures We also use Lewis symbols to indicate the formation of covalent bonds, which are shown in Lewis structures, drawings that describe the bonding in molecules and polyatomic ions. Answer b Because this element is located in Group 15, or 5A, on the periodic table, it will ionize to form an anion with a 3 charge. Compare the electron configurations of Rb. Krypton (Kr) is among the least reactive of all elements. Predict the charge on the stable ion formed by each of the following elements: (a) Be (b) P (c) F (d) S. The most stable monoatomic ion of bromine is Br-. The most common sulfur ion has a charge of: a) 2- b) 1- c) 4+ d) 6+ e) Sulfur does not form ions. 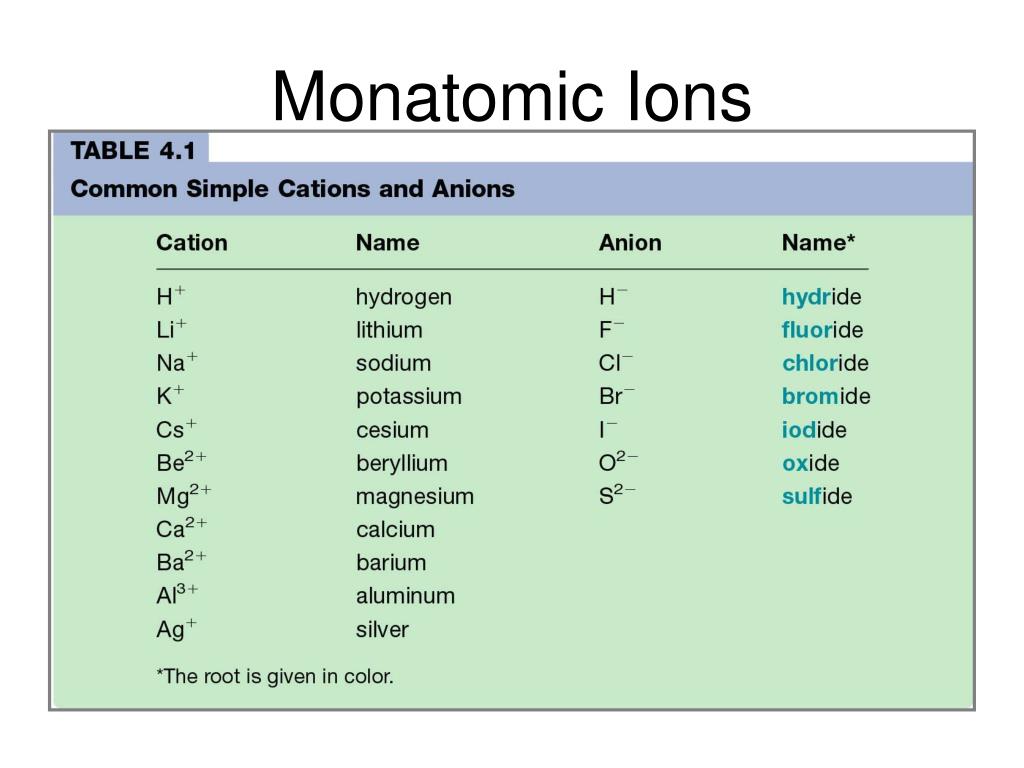
The suffix of the elements name is unmodified, because this ion is a cation. What is the most stable monatomic ion formed from caesium Which nuclide(s) would you predict to be stable (a) 48 K (b) 79 Br (c) argon 32 The only chemically stable ion of rubidium is Rb+. 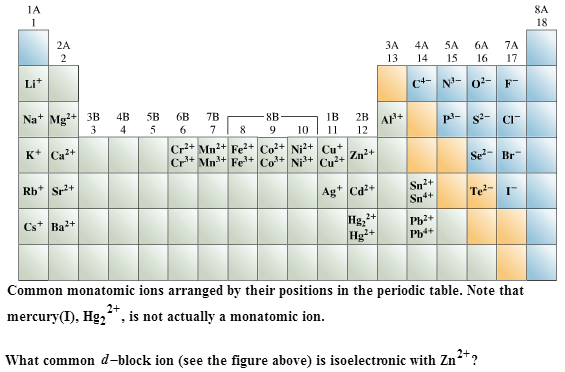
A monatomic ion (also called simple ion ) is an ion consisting of exactly one atom. The resultant ion is symbolized as Ba + 2 and is named the barium ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
